Factory source Diagnositc Kit For Follicle Stimulating Hormone - OEM China China Lansionbio CE ISO Approved Amh One Step Rapid Test Kit – Baysen
Factory source Diagnositc Kit For Follicle Stimulating Hormone - OEM China China Lansionbio CE ISO Approved Amh One Step Rapid Test Kit – Baysen Detail:
We believe in: Innovation is our soul and spirit. Excellent is our life. Shopper need to have is our God for OEM China China Lansionbio CE ISO Approved Amh One Step Rapid Test Kit, Our really specialized process eliminates the component failure and offers our shoppers unvarying top quality, allowing us to control cost, plan capacity and maintain consistent on time delivery.
We believe in: Innovation is our soul and spirit. Excellent is our life. Shopper need to have is our God for China Rapid Test Kit, Clinical Diagnostic Test Kit, Our faith is to be honest first, so we just supply high quality goods to our customers. Genuinely hope that we can be business partners. We believe that we can establish long time business relationship with each other. You can contact us freely for more information and pricelist of our products and solutions !
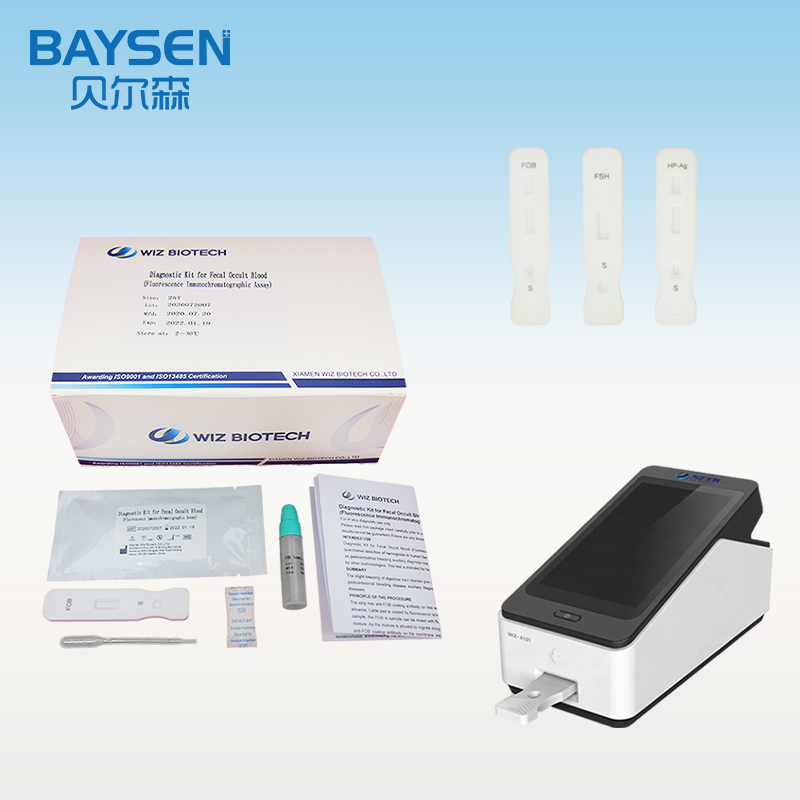
FOB Brochure



PRINCIPLE AND PROCEDURE OF FOB TEST
Princple:
The strip has anti-FOB coating antibody on test region, which is fastened to membrane chromatography in advance. Lable pad is coated by fluorescence labeled anti-FOB antibody in advance. When testing positive sample, the FOB in sample can be mixed with fluorescence labeled anti-FOB antibody, and form immune mixture. As the mixture is allowed to migrate along the test strip, the FOB conjugate complex is captured by anti-FOB coating antibody on the membrane and forms complex. The fluorescence intensity is positively correlated with the FOB content. The FOB in sample can be detected by fluorescence immunoassay analyzer.
Test Procedure:
1.Lay aside all reagents and samples to room temperature.
2.Open the Portable Immune Analyzer(WIZ-A101), enter the account password login according to the operation method of the instrument, and enter the detection interface.
3.Scan the dentification code to confirm the test item.
4.Take out the test card from the foil bag.
5.Insert the test card into the card slot, scan the QR code, and determine the test item.
6.Remove the cap from the sample tube and discard the first two drops diluted sample, add 3 drops (about 100uL) no bubble diluted sample verticaly and slowly into sample well of the card with provided dispette.
7.Click the “standard test” button, after 15 minutes, the instrument will automatically detect the test card, it can read the results from the display screen of the instrument, and record/print the test results.
8.Refer to the instruction of Portable Immune Analyzer(WIZ-A101).

You May like
SARS-CoV-2 Antigen Rapid Test(Colloidal Gold)
WIZ-A101 Portable Immune Analyzer
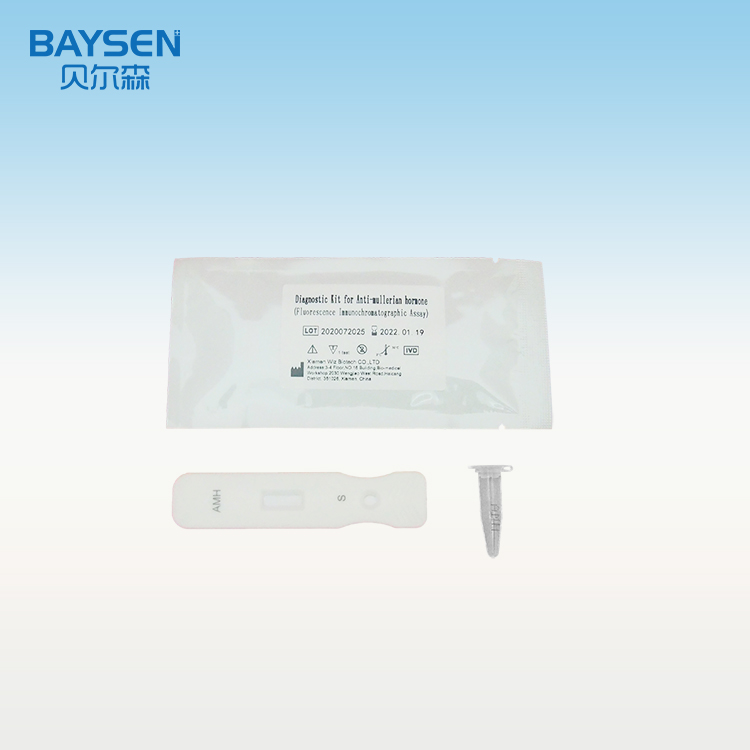
Diagnostic Kit for Anti-Mullerian Hormone (Fluorescence Immunochromatographic Assay)
About Us

Xiamen Baysen Medical Tech limited is a high biological enterprise which devotes itself to filed of fast diagnostic reagent and integrates research and development, production and sales into a whole. There are many advanced research staffs and sales managers in the company, all of them are have rich working experience in china and international biopharmaceutical enterprise.
Certificate display

Product detail pictures:


Related Product Guide:
Only Conservative Use of PSA Testing Is Cost-effective 3090D553-9492-4563-8681-AD288FA52ACE Group 2 34A8E98B-62ED-4216-98D6-E986304F4C2E | Diagnostic Kit For Isoenzyme Mb Of C Reatine Kinase
Commutability of the First World Health Organization International Standard for Human Cytomegalovirus | P24 Test Strips
Our organization promises all customers with the first-class products and solutions and the most satisfying post-sale service. We warmly welcome our regular and new clients to join us for Factory source Diagnositc Kit For Follicle Stimulating Hormone - OEM China China Lansionbio CE ISO Approved Amh One Step Rapid Test Kit – Baysen , The product will supply to all over the world, such as: Russia, Ethiopia, Philippines, After years' creating and developing, with the advantages of trained qualified talents and rich marketing experience, outstanding achievements were gradually made. We get good reputation from the customers due to our good solutions quality and fine after-sale service. We sincerely wish to create a more prosperous and flourishing future together with all the friends home and abroad!
Product variety is complete, good quality and inexpensive, the delivery is fast and transport is security, very good, we are happy to cooperate with a reputable company!














