Manufacturer of One Step Chlamydia Test - Diagnostic Kit(Colloidal Gold)for Human Chorionic Gonadotrophin – Baysen
Manufacturer of One Step Chlamydia Test - Diagnostic Kit(Colloidal Gold)for Human Chorionic Gonadotrophin – Baysen Detail:
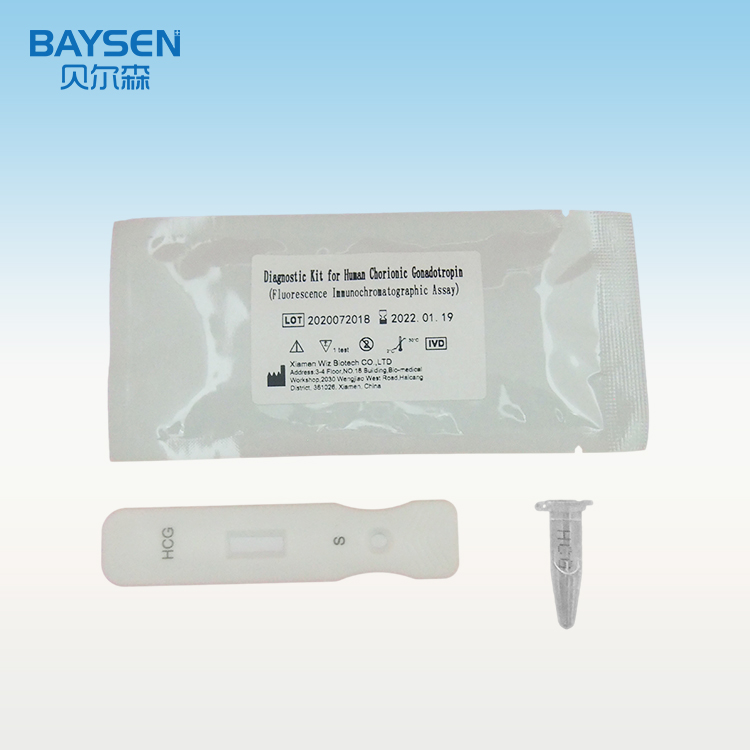
Diagnostic Kit(Colloidal Gold)for Human Chorionic Gonadotrophin
For in vitro diagnostic use only
Please read this package insert carefully prior to use and strictly follow the instructions. Reliability of assay results cannot be guaranteed if there are any deviations from the instructions in this package insert.
INTENDED USE
Diagnostic Kit(Colloidal Gold)for Human Chorionic Gonadotrophin is a colloidal gold immunochromatographic assay for the qualitative detection of human chorionic gonadotropin (HCG) levels in human serum and urine, it is used for early pregnancy diagnosis This test is a screening reagent. All positive sample must be confirmed by other methodologies. This test is intended for healthcare professional use only.
PACKAGE SIZE
1 kit /box, 10 kits /box, 25 kits,/box, 50 kits /box.
SUMMARY
HCG is a glycoprotein hormone secreted by the developing placenta after egg fertilization. HCG levels can be rapidly elevated in serum or urine as early as 1 to 2.5 weeks during pregnancy, and reach a peak in 8 week, than fall to medium level in 4 month, and maintained the level until the end of the pregnancy[1]. The Kit is a simple, visual qualitative test that detects HCG antigen in human serum or urine. The Diagnostic Kit is based on immunochromatography and can give a result within 15 minutes.
ASSAY PROCEDURE
1.Take out the test card from the foil bag, put it on the level table and mark it.
2.Discard the first two drops sample, add 3 drops (about 100μL) no bubble sample verticaly and slowly into sample well of the card with provided dispette, start timing.
3.The result should be read within 10-15 minutes, and it is invalid after 15 minutes.
Product detail pictures:

Related Product Guide:
Een kudtkoekiewall. Omdat dat moet, van de kudtkoekiewet. | P24 Test Strips
Many older men still getting PSA test for prostate cancer, against recommendations | Calprotectin Elisa Kit
We keep on with the theory of "quality first, provider initially, constant improvement and innovation to meet the customers" with the management and "zero defect, zero complaints" as the standard objective. To great our company, we deliver the merchandise using the fantastic excellent at the reasonable price for Manufacturer of One Step Chlamydia Test - Diagnostic Kit(Colloidal Gold)for Human Chorionic Gonadotrophin – Baysen , The product will supply to all over the world, such as: Luxembourg, Bolivia, Russia, Our team knows well the market demands in different countries, and is capable of supplying suitable quality products and solutions at the best prices to different markets. Our company has already set up a experienced, creative and responsible team to develop clients with the multi-win principle.
We have been cooperated with this company for many years, the company always ensure timely delivery ,good quality and correct number, we are good partners.