OEM Manufacturer Clinical Chemistry Analyzer - family laymen use antigen nasal rapid test for covid-19 – Baysen
OEM Manufacturer Clinical Chemistry Analyzer - family laymen use antigen nasal rapid test for covid-19 – Baysen Detail:
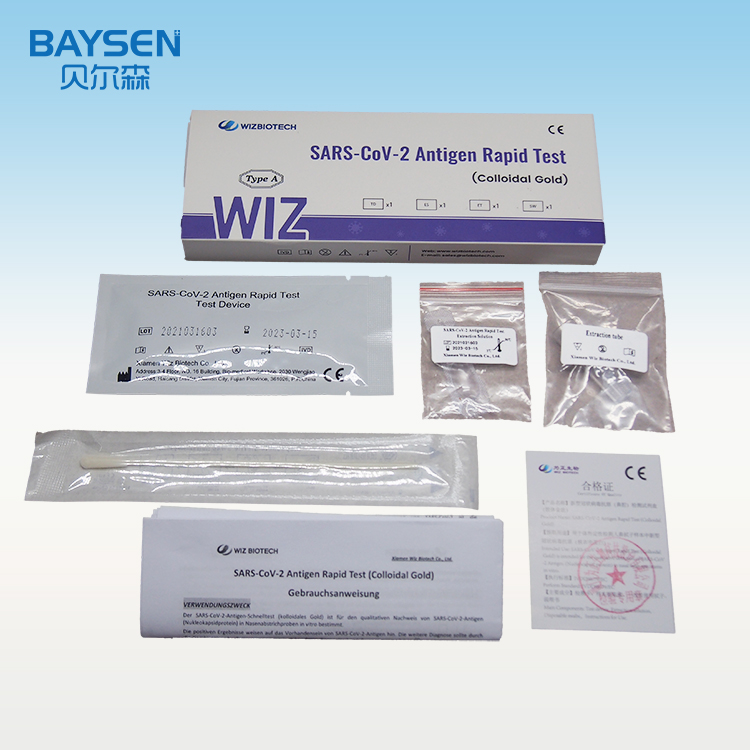
SARS-CoV-2 Antigen Rapid Test (Colloidal Gold) is intended for the qualitative detection of SARS-CoV-2 Antigen (Nucleocapsid protein) in nasal swab specimens in vitro.
ASSAY PROCEDURE
Before using the reagent, operate it strictly according to the Instruction for Use to ensure the accuracy of the results.
1. Before the detection, the test device and the sample are taken out from the storage condition and balanced to room temperature (15-30℃).
2. Tearing the packaging of the aluminum foil pouch, take out the test device, and place it horizontally on the test table.
3. Vertically invert the specimen extraction tube (the extraction tube with processed specimens), add 2 drops vertically into the sample well of the test device.
4. The test results should be interpreted within 15 to 20 minutes, invalid If more than 30 minutes.
5. Visual interpretation can be used in result interpretation.
Product detail pictures:

Related Product Guide:
Why I Won’t Get a PSA Test for Prostate Cancer | P24 Test Strips
PSA testing for prostate cancer | Health Information | P24 Test Strips
We've numerous great employees customers excellent at promoting, QC, and working with kinds of troublesome difficulty inside the generation method for OEM Manufacturer Clinical Chemistry Analyzer - family laymen use antigen nasal rapid test for covid-19 – Baysen , The product will supply to all over the world, such as: Singapore, Florence, Istanbul, We have the best products and professional sales and technical team.With the development of our company, we are able to provide customers best products, good technical support, perfect after-sales service.
We have worked with many companies, but this time is the best,detailed explanation, timely delivery and quality qualified, nice!