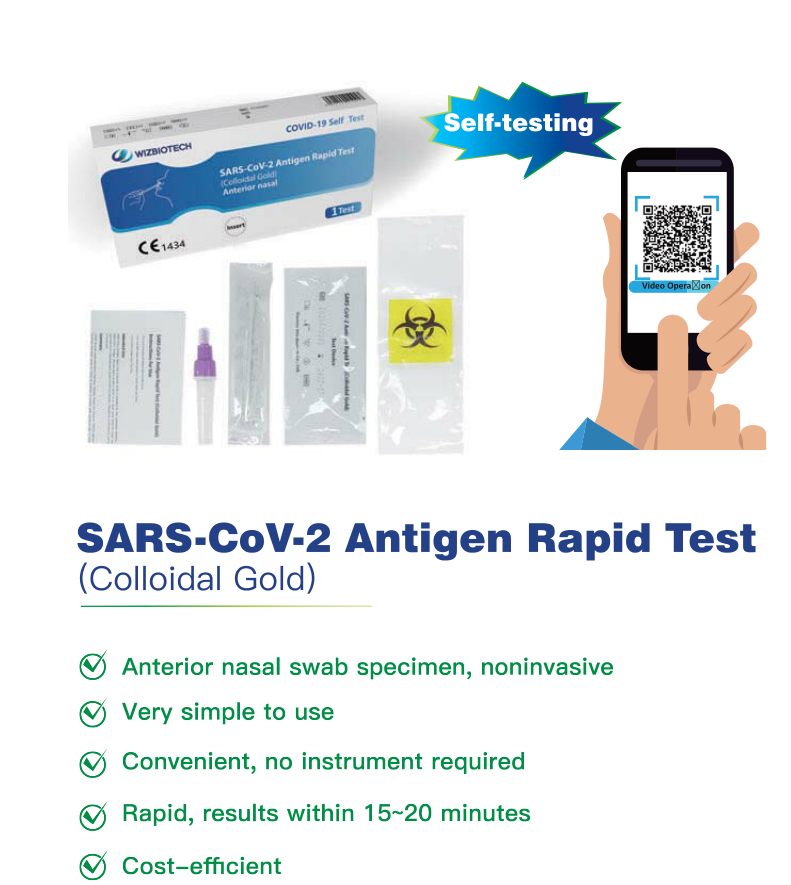
I-Mylasia ivumile ukuthi i-SARS-CoV-2 antigen test kit izihlolele yona ngokwayo.
I-Mylasia ivumile ukuthi i-SARS-CoV-2 antigen test kit izihlolele yona ngokwayo.
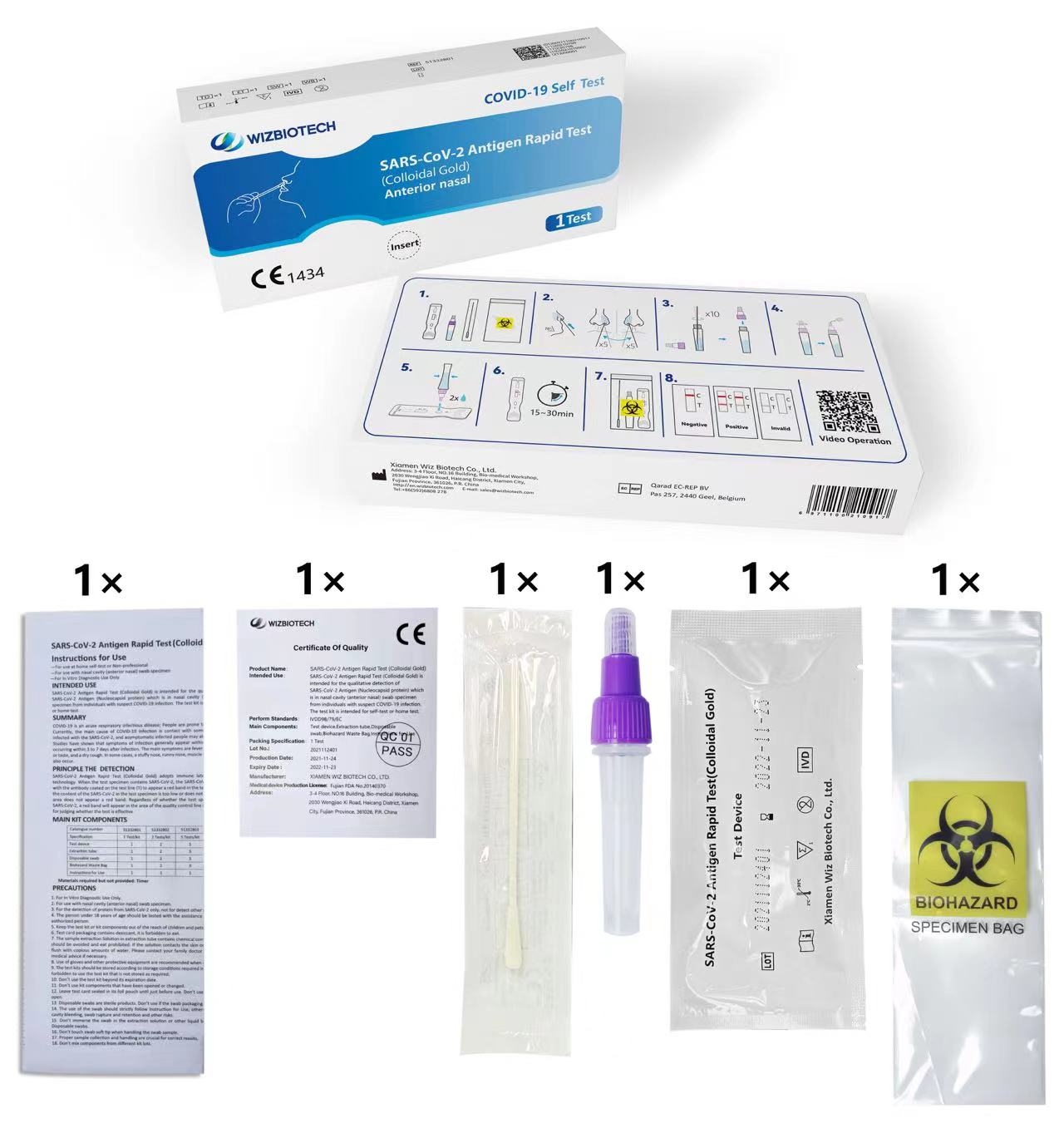
Imiyalelo Yokusebenzisa
—Kungasetshenziswa ekhaya
ukuzihlola noma okungeyona ingcweti
—Kusetshenziswa nesampula yeswabhu yekhala (ingaphambili lekhala)
—Ukusetshenziswa Kokuxilongwa Kwe-In Vitro Kuphela
Isitoreji
Ikhithi yokuhlola kufanele igcinwe endaweni epholile engu-2°C ~ 30°C, yomile futhi ingangeni elangeni eliqondile (Ungayiqandisi ikhithi noma izingxenye zayo).
Isikhathi sokuphila seshelufu sekhithi siyizinyanga eziyi-12.
Ikhadi lokuhlola kufanele lisetshenziswe zingakapheli imizuzu engama-60 ngemva kokuvula isikhwama se-aluminium foil.
Ukuze uthole usuku lokuphelelwa yisikhathi kwekhithi, sicela ubheke ilebula lomkhiqizo.
Ukuzwela:98.26%(95%CI 93.86%~99.79%)
Ukucaciswa:100.00%(95%CI 99.19%~100.00%)
Inani Elibikezelayo Elihle:100%(95%CI 96.79%~100.00%)
Inani Lokubikezela Elibi: 99.56% (95%CI 98.43% ~ 99.95%)
Isivumelwano Sephesenti Eliphelele: 99.65% (95% CI 98.74~99.96%)
Ukuhlolwa Okusheshayo Kwe-SARS-CoV-2 Antigen kuhloselwe ukuthola i-SARS-CoV-2 Antigen esezingeni eliphezulu ku-oropharyngea swab kanye ne-nasopharyngeal swab specimens in Vitro