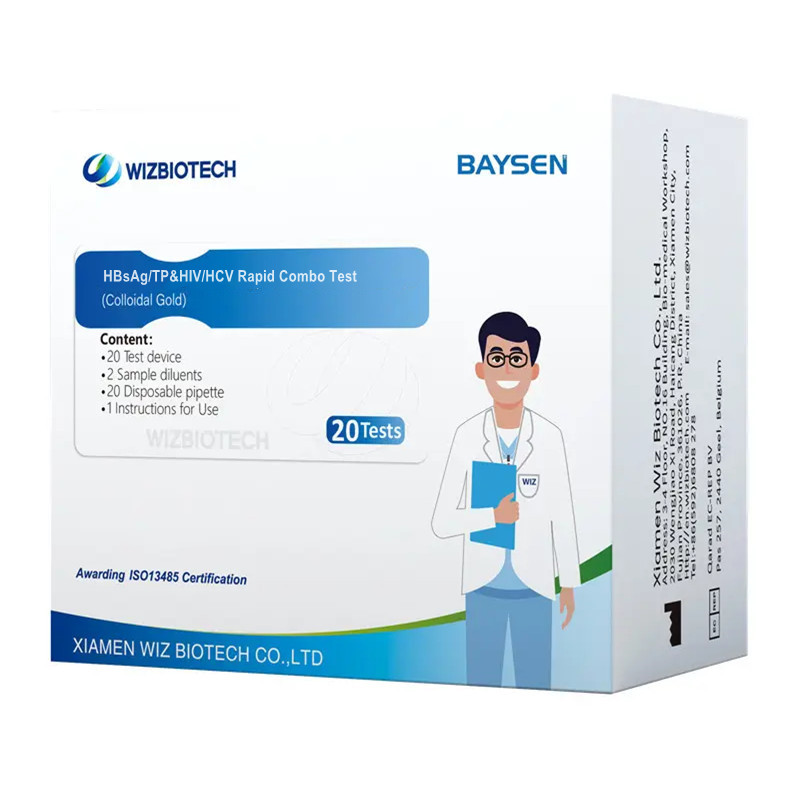
Uvavanyo lwe-HCV HBSAG kunye neSyphilish Rapid Combo Test
ULWAZI LWEMVELISO
| Inombolo yoMzekelo | I-HBsAg/TP kunye ne-HIV/HCV | Ukupakisha | Iimvavanyo ezingama-20/ikhithi, iikhithi ezingama-30/i-CTN |
| Igama | Uvavanyo lwe-HBsAg/TP kunye ne-HIV/HCV olukhawulezayo | Ukuhlelwa kwezixhobo | Iklasi yesi-3 |
| Iimbonakalo | Uvakalelo oluphezulu, Ukusebenza lula | Isatifikethi | I-CE/ ISO13485 |
| Ukuchaneka | > 97% | Beka ubomi kwishelufa | Iminyaka emibini |
| Indlela yokusebenza | Igolide yeColloidal | Inkonzo ye-OEM/ODM | Iyafumaneka |
Ukubaluleka
Ixesha lokuvavanya: 15-20mins
Indawo yokugcina: 2-30℃/36-86℉
Indlela yokusebenza: Igolide yeColloidal
Uphawu:
• Uvakalelo oluphezulu
• ukufundwa kweziphumo kwimizuzu eli-15-20
• Ukusebenza okulula
• Ukuchaneka Okuphezulu
UKUSETYENZISWA OKUJOLISWEYO
Le khithi ifanelekile ekumiselweni kwe-in vitro qualitative virus ye-hepatitis B, i-syphilis spirochete, i-human immunodeficiency virus, kunye ne-hepatitis C virus kwi-human serum/plasma.iisampuli zegazi/igazi elipheleleyo ukuze kufunyanwe isifo se-hepatitis B, i-syphilis spirochete, i-human immunodeficiency virus, kunye nosulelo lwe-hepatitis C. Iziphumo ezifunyenweyo kufunekaIhlalutywe kunye nolunye ulwazi lwezonyango. Yenzelwe ukusetyenziswa ziingcali zonyango kuphela.
Inkqubo yovavanyo
| 1 | Funda imiyalelo yokusetyenziswa kwaye ulandelane ngokungqongqo nemiyalelo yokusetyenziswa efunekayo ukuze uphephe ukuphazamisa ukuchaneka kweziphumo zovavanyo. |
| 2 | Ngaphambi kovavanyo, ikhithi kunye nesampulu zithathwa kwindawo yokugcina kwaye zilinganiswe kubushushu begumbi kwaye ziphawulwe. |
| 3 | Ukrazula iphakheji yesikhwama sefoyile ye-aluminium, khupha isixhobo sovavanyo usiphawule, uze usibeke ngokuthe tye etafileni yovavanyo. |
| 4 | Faka iisampulu ze-serum/plasma nge-dropper elahlwayo uze wongeze amaconsi ama-2 kwiqula ngalinye le-s1 kunye ne-s2; yongeza amaconsi ama-3 kwiqula ngalinye le-s1 kunye ne-s2 kwisampulu yegazi elipheleleyo ngaphambi kokongeza amaconsi ama-1-2 esicombululo sokuhlamba kwiqula ngalinye le-s1 kunye ne-s2 uze uqalise ixesha. |
| 5 | Iziphumo zovavanyo mazitolikwe kwimizuzu eli-15 ukuya kwengama-20, ukuba iziphumo ezitolikwe ngaphezulu kwemizuzu engama-20 azivumelekanga. |
| 6 | Ukutolika okubonakalayo kunokusetyenziswa ekutolikeni iziphumo. |
Qaphela: isampulu nganye mayifakwe ngepayipi ecocekileyo elahlwayo ukuze kuthintelwe ungcoliseko oluhambelanayo.
UKUSEBENZA KWEENKLINIKI
| Iziphumo zeWIZ zeI-HBsag
| Iziphumo zovavanyo lwe-reagent yesalathiso | Izinga elifanelekileyo lokungaqondani: 99.06% (95%CI 96.64%~99.74%) Izinga lokungaqondani elibi: 98.69% (95%CI96.68%~99.49%) Izinga elipheleleyo lokungaqondani: 98.84% (95%CI97.50%~99.47% | ||
| Elungileyo | Okungalunganga | Isimbuku | ||
| Intle | 211 | 4 | 215 | |
| Okungalunganga | 2 | 301 | 303 | |
| Isimbuku | 213 | 305 | 518 | |
| Iziphumo zeWIZ zeTP
| Iziphumo zovavanyo lwe-reagent yesalathiso | Izinga elifanelekileyo lokungaqondani: 96.18% (95%CI 91.38%~98.36%) Izinga lokungaqondani elibi: 97.67% (95%CI95.64%~98.77%) Izinga elipheleleyo lokungaqondani: 97.30% (95%CI95.51%~98.38%) | ||
| Elungileyo | Okungalunganga | Isimbuku | ||
| Intle | 126 | 9 | 135 | |
| Okungalunganga | 5 | 378 | 383 | |
| Isimbuku | 131 | 387 | 518 | |
| Iziphumo zeWIZ zeI-HCV
| Iziphumo zovavanyo lwe-reagent yesalathiso | Izinga elifanelekileyo lokungaqondani: 93.44% (95%CI 84.32%~97.42%) Izinga lokungaqondani elibi: 99.56% (95%CI98.42%~99.88%) Izinga elipheleleyo lokungaqondani: 98.84% (95%CI97.50%~99.47%) | ||
| Elungileyo | Okungalunganga | Isimbuku | ||
| Intle | 57 | 2 | 59 | |
| Okungalunganga | 4 | 455 | 459 | |
| Isimbuku | 61 | 457 | 518 | |
| Iziphumo zeWIZ zeI-HIV
| Iziphumo zovavanyo lwe-reagent yesalathiso | Izinga elifanelekileyo lokungaqondani: 96.81% (95%CI 91.03%~98.91%) Izinga lokungaqondani elibi: 99.76% (95%CI98.68%~99.96%) Izinga elipheleleyo lokungaqondani: 99.23% (95%CI98.03%~99.70%) | ||
| Elungileyo | Okungalunganga | Isimbuku | ||
| Intle | 91 | 1 | 92 | |
| Okungalunganga | 3 | 423 | 446 | |
| Isimbuku | 94 | 424 | 518 | |